 
49.5% x 195 / 12 = 8.0 mol carbon (2x amount in empirical calculations).Find empirical/molecular formula of caffeine, which contains 49.5% carbon, 5.15% hydrogen, 28.9% nitrogen, 16.5% oxygen.combustion analysis - all the C goes into CO 2 and all the H goes into H 2O in combustion moles of C/H in original compound can be found from the masses of CO 2/H 2O in the product.whole number multiple can be found by dividing the molecular weight by empirical formula weight.subscripts in the molecular formula can be found by multiplying the subscripts in the empirical formula by a whole number.C 6H 12O 2 (molecular formula) > CH 2O (empirical formula).finding the ratios of elements in a compound gives the empirical formula (from percentage composition).How many particles of carbon-12 are found in 12 grams of it? 12 g / 12/g/mol x 6.02 x 10 23 = 6.02 x 10 23Įmpirical formula - tells relative number of atoms of each element in a compound.divide the mass by the representative particle's molar mass and then multiply it by 6.02 x 10 23.Mass (grams ) to number of representative particles divide number of representative particles by 6.02 x 10 23 and then multiply that by the representative particle's molar mass.Number of representative particles to mass (grams ) How many grams are in 23 moles of iron? 23 x 55.845 = 1.3 x 10 3.multiply number of moles by the representative particle's molar mass.How many moles are in 24 grams of carbon-12? 24/12 = 2.divide the mass by the representative particle's molar mass.divide number of representative particles by 6.02 x 10 23.Number of representative particles to moles How many carbon particles in four moles of carbon? 4 x 6.02 x 10 23 = 2.408 x 10 24.multiply number of moles by 6.02 x 10 23. 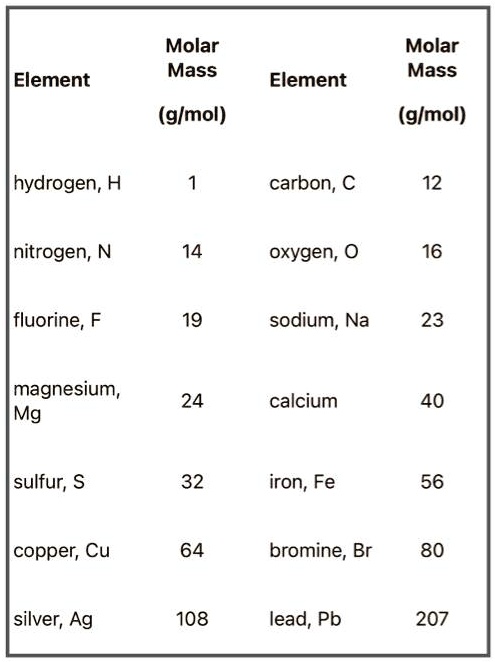 
Moles to number of representative particles use dimensional analysis to convert between masses, moles, and numbers of particles.molar mass - numerically equal to the element's atomic mass (1 atom of carbon-12 = 12 amu, 1 mol of carbon-12 = 12 grams).Avogadro's number (mole) - number of atoms in 12g of pure carbon-12 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
